 It’s a battle fought by all living things. Only the individuals that are best suited to the world in which they live have a chance of surviving: what we know as survival of the fittest. And at no point in these babies’ lives is this more critical than when they leave the nest. Hungry predators are waiting. Although they may look the same, each baby is different from the next. The tiniest variation in size and shape can determine who lives and who dies. Some are weak, others strong. Anything that boosts the baby’s chances of surviving, such as a sturdier shell or longer flippers, will be passed on to its young in its genes and over time these inherited changes can lead to the evolution of a new species. These babies are the genetic veterans of a battle for survival that has been going on since the first turtles evolved. Here only a few make it to the water’s edge. This is natural selection, the way life evolves, and it has been shaping new kinds of creatures throughout the whole of life’s great journey.
It’s a battle fought by all living things. Only the individuals that are best suited to the world in which they live have a chance of surviving: what we know as survival of the fittest. And at no point in these babies’ lives is this more critical than when they leave the nest. Hungry predators are waiting. Although they may look the same, each baby is different from the next. The tiniest variation in size and shape can determine who lives and who dies. Some are weak, others strong. Anything that boosts the baby’s chances of surviving, such as a sturdier shell or longer flippers, will be passed on to its young in its genes and over time these inherited changes can lead to the evolution of a new species. These babies are the genetic veterans of a battle for survival that has been going on since the first turtles evolved. Here only a few make it to the water’s edge. This is natural selection, the way life evolves, and it has been shaping new kinds of creatures throughout the whole of life’s great journey.So runs the undoubting, modern account of how living things come to be. Whether true or false, it is, properly speaking, a myth: an imaginative story to help explain the world as it has become. The fossil record itself tells us nothing about the processes giving rise to novel forms, and there’s no reason to think that modern turtles are better adapted to their environments than their predecessors were to theirs. Survival rates remain extremely low, as the footage of hungry pelicans swooping down on the toddlers brutally emphasises.
Like the extinct ichthyosaurs, turtles (including tortoises and terrapins) are reptiles, and they have terrestrial ancestors. Why they should have wanted to swap the land for the sea remains unclear, for the sea was a hostile environment, and they needed ‘adaptations’ to cope with it before venturing forth. Even today, sea turtles must draw their oxygen from the air and come ashore to lay their eggs. Whatever its powers, natural selection has not delivered them from these handicaps. And surely it must be a handicap that so few progeny make it to adulthood (perhaps 1 in 1000). Ichthyosaurs, which evolved from terrestrial reptiles about the same time as turtles, also had to draw their oxygen from the air, but they gave birth to live young, in the sea. They went extinct, eventually; turtles did not. It is as if an invisible Designer wished to show that he could solve the problem of reproduction either way, and survival was not contingent on the solution.
Perhaps the best known sea turtle is the leatherback. Guided by a mysterious instinct that one would struggle to reduce to genetics, the pregnant animal emerges from the surf after dark, close to high tide. Laboriously she crawls up the beach, selects a site just beyond the high-water mark and starts to dig. Her front flippers scoop out a cavity for her body, the rear flippers excavating a smaller, deeper hole. Typically she deposits around 70 fertile eggs and 40 smaller infertile ones. Her duty done, she fills the nest with sand to conceal its location and returns to the sea. She will never know her infants.
She repeats the burial ritual several times in the course of the nesting season. In about two months, that first clutch will hatch, and over a few days the newborn turtles will dig their way to the surface. Like their unseen mothers, they know what to do by instinct. Predators may be lurking, and the babies sense that they must wait for the top sand layers to cool – a sign of nightfall – before making their break for the sea.
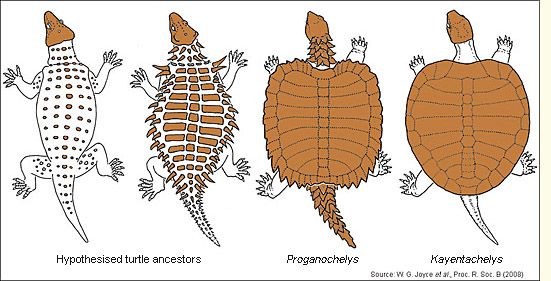
 Turtle fossils in the Triassic are rare, but by the end of the Triassic they occur globally. The oldest goes back to the Middle Triassic, just before the earliest dinosaurs. Among the best known, from the Late Triassic, is Proganochelys, found in Thailand and in several parts of Germany, where the sedimentary clues suggest a brackish or marginal marine environment. The turtle may have been washed down to its resting place by coastal rivers. Its forelimb dimensions and heavy armour point to a terrestrial existence (Joyce & Gauthier 2003), but the ratio of the width of carapace (dorsal shell) to plastron (bony underbelly) suggest it was aquatic (Lichtig & Lucas 2017). It had toed feet, teeth on the palate (but not on the jaws), a spiky neck with ribs, and a club-like tail. If the tail was used for defence, it would have had little effect in water. The teeth hinted at an ancestry among non-turtles (modern turtles do not have teeth).
Turtle fossils in the Triassic are rare, but by the end of the Triassic they occur globally. The oldest goes back to the Middle Triassic, just before the earliest dinosaurs. Among the best known, from the Late Triassic, is Proganochelys, found in Thailand and in several parts of Germany, where the sedimentary clues suggest a brackish or marginal marine environment. The turtle may have been washed down to its resting place by coastal rivers. Its forelimb dimensions and heavy armour point to a terrestrial existence (Joyce & Gauthier 2003), but the ratio of the width of carapace (dorsal shell) to plastron (bony underbelly) suggest it was aquatic (Lichtig & Lucas 2017). It had toed feet, teeth on the palate (but not on the jaws), a spiky neck with ribs, and a club-like tail. If the tail was used for defence, it would have had little effect in water. The teeth hinted at an ancestry among non-turtles (modern turtles do not have teeth).
Another key fossil is Odontochelys, from China. Odontochelys is both slightly older and in some respects less evolved than Proganochelys – for example, it had teeth on both upper and lower jaws, its overall shape was distinctly elongate and it lacked a fully developed carapace, as if development of this part of the anatomy were arrested at the embryonic stage (a phenomenon known as heterochrony); the dorsal ribs were nonetheless broadened and flattened. As in some freshwater turtles today, the carapace  may have consisted of non-bony tissue and so not been preserved. The habitat of Odontochelys was probably river delta or shallow marine. Eorhynchochelys was found at the same site 7.5 m lower in the rocks and was thus slightly older. It had neither carapace nor plastron, and had a long tail. On the other hand, it had a beak (an advanced feature) and lacked teeth. Its habitat could not be determined.
may have consisted of non-bony tissue and so not been preserved. The habitat of Odontochelys was probably river delta or shallow marine. Eorhynchochelys was found at the same site 7.5 m lower in the rocks and was thus slightly older. It had neither carapace nor plastron, and had a long tail. On the other hand, it had a beak (an advanced feature) and lacked teeth. Its habitat could not be determined.
Thus, in quick succession, we have a turtle with no plastron or carapace, a soft-shelled turtle and a hard-shelled turtle. However, it would be a mistake to see them as forming a simple evolutionary succession. They differ from each other in many respects, some features come and go, and the juvenile appearance of the soft shell of Odontochelys suggests that the genetic program for a carapace may already have been present. At least some of the Upper Triassic turtles were terrestrial; some were probably also comfortable in rivers, or even shallow coastal environments. The carapace was not an adaptation to life in the sea.
Turtles after the Triassic split into two main groups, the ‘pleurodires’ and the ‘cryptodires’. Although neither sub-group initially had the neck retraction mechanisms that would become their most obvious characteristic, pleurodires (‘side-necks’) later folded their necks to the side of the shell, while cryptodires (‘hidden-necks’) retracted the head by pulling their necks up and back. Both mechanisms require complex, co-ordinated modifications of the neck vertebrae and muscles. Living turtles are either pleurodires or cryptodires. Triassic turtles protected their necks by other means: Proganochelys by a collar of horny spikes, Palaeochersis (another Late Triassic turtle) by an extension of the carapace.
 Pleurodires are now the less common group, restricted to the continents of the southern hemisphere, but once they were very widespread, both on land and in the estuaries and shallow seas around the coasts. Sea turtles (Chelonioidea) and soft-shell turtles (Trionychoidea) are all cryptodires.
Pleurodires are now the less common group, restricted to the continents of the southern hemisphere, but once they were very widespread, both on land and in the estuaries and shallow seas around the coasts. Sea turtles (Chelonioidea) and soft-shell turtles (Trionychoidea) are all cryptodires.
The oldest fossil sea turtle is Santanachelys, from the Early Cretaceous. Like its modern counterparts, it had huge salt glands under the eyes, essential for excreting the salts that accumulated as a result of living off seafood. The metacarpals and short digits of the feet were still moveable, as in land turtles: only later did the toe bones lengthen and become encased in flesh, and the feet turn into flippers. As the Cretaceous progressed, sea-turtle diversity exploded. The largest animal grew to a width of 4 metres and a length of 6 metres. Some flippers were lengthened by increasing the number of toe bones.
 Towards the end of the period, amazingly, some of the cryptodires began an evolutionary journey back to the land. Present-day tortoises (Testudinidae) descend from turtles that swam in the sea, not directly from older land-going turtles. The anatomical changes involved are not well documented, but many turtles again became toe-walkers, and while the number of scutes (scales on the carapace) remained constant, shell ornamentation and shape varied greatly. The oldest tortoise fossils come from the late Palaeocene of Mongolia. By then the transition back to the land, at least in one lineage, was already complete. In the Eocene, tortoises colonised North America, Europe and Africa and adapted to an immense range of environments, from forests to deserts. Some were capable of subsisting both in water and on land.
Towards the end of the period, amazingly, some of the cryptodires began an evolutionary journey back to the land. Present-day tortoises (Testudinidae) descend from turtles that swam in the sea, not directly from older land-going turtles. The anatomical changes involved are not well documented, but many turtles again became toe-walkers, and while the number of scutes (scales on the carapace) remained constant, shell ornamentation and shape varied greatly. The oldest tortoise fossils come from the late Palaeocene of Mongolia. By then the transition back to the land, at least in one lineage, was already complete. In the Eocene, tortoises colonised North America, Europe and Africa and adapted to an immense range of environments, from forests to deserts. Some were capable of subsisting both in water and on land.
They also colonised some islands. The Galapagos islands are named after these animals, their carapaces reminding Spanish explorers of a kind of saddle they called a ‘galapago’. Their closest relatives are a species endemic to Chile, 1,000 kilometres to the east. Since the archipelago is volcanic in origin and has never been connected to the mainland, they must have reached the islands by rafting on tree branches as currents transported them up the coast of Chile and Peru before circulating westward. Giant tortoises used to exist on all continents, but except on the oceanic islands of the Galapagos and the Seychelles, they are now extinct. The largest, Colossochelys atlas, attained the size of a Volkswagen Beetle.
Today sea turtles are much less diverse than in prehistoric times: just two families and six genera. The leatherback is a family all by itself. As its name implies, it differs from the others in having a soft, cartilaginous shell rather than a hard bony one. It also differs in having no scutes, a reduced skeleton, a throat and upper digestive tract lined with spines (to stop swallowed jellyfish from escaping) and an ability to generate some of its body heat from within. The other marine family comprises the Kemp’s ridley and olive ridley, the green and black sea turtles, the loggerhead, the hawksbill, and the flatback. Most of these genera are capable of hybridising with others in the family (Bowen & Karl 1997).
Modern turtles have sufficient features in common to substantiate the view that they had a common ancestor. Recent finds have suggested that that ancestor must have been a reptile with two temporal fenestrae, and that these were subsequently lost. In Eorhynchochelys the upper fenestra was closed and the lower fenestra partially closed. The slightly later Proganochelys and Odontochelys were,, like modern turtles, already anapsids.
The evolutionary predecessors of the Triassic forms are assumed not to have been recognisably turtles at all. That seems likely, though not certain. One suggestion was that turtles derived from a group of Permian anapsids called the pareiasaurs, the smallest of which showed ‘otherwise uniquely turtle features such as a rigid covering of dermal armour over the entire dorsal region, expanded flattened ribs, cylindrical scapula blade, great reduction of humeral torsion (to 25°), greatly developed trochanter major [an eminence near the top of the femur], offset femoral head, and reduced cnemial crest [at the top] of the tibia’ (Lee 1997). Plausible though the hypothesis appeared at the time, it has since been rejected by most specialists (e.g. Rieppel & Reisz 1999). One reason is that the carapace or ‘dermal armour’ of turtles is a complex structure formed through the fusion of the vertebrae and ribs to interlocking plates beneath the skin and through the growth of horny scutes above the skin; as such it is without parallel. The dermal plates of pareiasaurs cannot be interpreted as precursors of the plates in the turtle carapace. Furthermore, the limb girdles and shoulder blades lie within the ribcage rather than outside it as in other amniotes, and if the turtle ancestor had been a terrestrial animal, the ribs would somehow have had to lose their function of supporting respiration and locomotion:
In a generalized tetrapod reptile, aspiration of air is effected by an expansion of the body cavity through muscular action exerted on the ribs. Exhalation is effected either by passive recoil of the body walls, and/or by compression of the lungs as a result of active compression of the rib cage. By contrast, respiration in turtles depends on volume changes of the thoracico-peritoneal cavity inside the rigid dermal armor, which is achieved by altering the position of the limb flanks through the activity of anterior and posterior muscles. A comparison of respiration in an aquatic (Chelydra) and a terrestrial (Testudo) turtle resulted in the identification of three forces that influence the relative volume of the thoracico-peritoneal cavity: These forces are muscular activity, gravity (supporting inhalation), and, in aquatic turtles, hydrostatic pressure (supporting exhalation).
A terrestrial ancestor could not could not have relied on water pressure to mitigate its breathing problems. In short, the postulated origin from pareiasaurs ‘greatly oversimplified the evolutionary changes that took place in the origin of the turtle body plan’.
On the basis that turtles have to be placed somewhere in the presumed universal tree of life, the genetic evidence favours crocodiles as the closest living relatives. Crocodiles are diapsids: they have two pairs of fenestrae in the skull. However, a turtle + crocodilian grouping is ‘completely unorthodox from a morphological and paleontological perspective’ (Zardoya & Meyer 2001).
Turtle morphology is surrounded on all sides by profound discontinuity. ‘Due to a general lack of shared derived characters with other amniotes and due to conflicting phylogenetic signal in different data sets,’ it is not as yet possible to identify an ancestral group.
Nonetheless, turtles have evolved a great deal in the course of their fossil history. As Darwin perceived while visiting the Galapagos Islands, evolution happens when organisms colonise vacant ecospace: they adapt. Darwin speculated that they adapted through one variation being fortuitously more advantageous in the new environment than another. What specified an organism’s form in the first place was, at that time, a ‘black box’. Within a framework of philosophical materialism it was perhaps not unreasonable to suppose that variants came about randomly, that the favouring of one form over another was a hit-and-miss process of ‘mutate or die’. With the discovery of DNA, however, that view became suspect. Once it was known that the growing embryo made proteins and organised cells by following a set of instructions, the possibility opened up that evolutionary change too might be genetically controlled. DNA might determine phylogeny as well as ontogeny.
- the same modifications of shoulder girdle and limb extremities in several lines of marine and freshwater turtles (Depecker et al. 2006)
- the formation of paddles in each of the three chelonioid families (Hirayama 1998)
- very similar skull shapes, neck and skull colour patterns and shell shapes in the Emydidae, Geoemydidae and Testudinidae families (Claude et al. 2005)
- the re-appearance of a club-like tail, 180 million years after Proganochelys, in the tortoise Meiolania
- the re-appearance of a pelvis sutured to the shell in pleurodires 50 million years after the Late Triassic Proterochersis (Joyce et al. 2013)

Another example is the innovation of salt glands in groups as diverse as sharks, estuarine crocodiles, sea snakes, marine birds and marine iguanas. Just what is involved in the innovation may be gathered from this description:
Sea turtle salt glands are modified lachrymal glands, each gland consisting of about one hundred lobules separated by blood vessels and connective tissue. The lobules contain many blind-ending secretory tubules which drain into a central canal within each lobule, and the central canals combine to form secondary ducts and a sac-like main duct which opens into the posterior canthus. The central canal and secretory tubules have a rich blood supply and there is extensive vascularization of the duct. The central canals are surrounded by broad sheaths of connective tissue with a rich network of cholinesterase staining nerve fibers around each tubule; a nerve network reactive to monoamine oxidase is also found in the perilobular connective tissue. The lobule is composed of three types of cell.
P. Lutz & J. A. Musick, 1996. The Biology of Sea Turtles, p 347.
If ascribing such intricate mechanisms to the work of chance is not the most plausible of interpretations, ascribing to coincidence the multiple re-occurrence of them compounds the difficulty. Even so simple a recurrent device as the egg-tooth, the ephemeral spike with which the baby reptile or bird instinctively hacks through the egg, cries out for a design explanation.
Amongst the other wonders that came into operation in the course of turtle evolution two more deserve mention.
The long-distance migrations of sea turtles represent some of the most remarkable feats of orientation and navigation in the animal kingdom. Starting from their birth places on the eastern coast of North America, juvenile loggerheads follow the Gulf Stream from one side of the North Atlantic to the other and back again, travelling distances of more than 9,000 miles (15,000 kilometres). In the Pacific the migrations can be even longer. Having deposited her eggs on a beach in Papua New Guinea, one adult loggerhead was tracked all the way to feeding grounds off the coast of Oregon: a distance of 12,774 miles. Nor, probably, was that the first time she had crossed the ocean. When the time comes to breed, loggerheads return to the beach where they were born. Somehow they remember its location by reference to the Earth’s magnetic field and navigate home with the precision of homing pigeons – which have miraculously ‘evolved’ a very similar system.
 Both ancestrally and as individuals, sea turtles originate on land, yet their terrestrial origin imposes no limit on their world. Like the ichthyosaurs, they have acquired an ability to live far out in the ocean and an ability to dive deep into it. Leatherbacks have been known to plumb depths of over 1,000 metres. though that far down they would have to depend on taste rather than sight. They gradually collapse their lungs as they dive, reducing their buoyancy, and draw the oxygen they need from cells rich in myoglobin and haemoglobin. Green sea turtles can stay under water for as long as five hours.
Both ancestrally and as individuals, sea turtles originate on land, yet their terrestrial origin imposes no limit on their world. Like the ichthyosaurs, they have acquired an ability to live far out in the ocean and an ability to dive deep into it. Leatherbacks have been known to plumb depths of over 1,000 metres. though that far down they would have to depend on taste rather than sight. They gradually collapse their lungs as they dive, reducing their buoyancy, and draw the oxygen they need from cells rich in myoglobin and haemoglobin. Green sea turtles can stay under water for as long as five hours.
Like all reptiles, turtles need to absorb heat from their environment in order to keep warm. Ostensibly it was not a wise move for land turtles to try to make their living in the ocean, where temperatures were lower than on land. Special equipment was needed. But as we have seen, special equipment was just what they acquired. To conserve energy they have relatively low rates of metabolism. Leatherbacks expose themselves to colder temperatures than any other turtle but are also more active, an instinct for constant movement causing them to generate high levels of heat in their muscle tissues. A thick layer of fat under the carapace provides insulation, while larger body size, and consequently a greater volume-to-surface ratio, further reduces the heat loss. The flippers have a low volume-to-surface ratio, but this is counteracted by arranging the arteries and veins next to each other and wrapping them in insulating fibre, so that heat from the blood flowing out is transferred to the cooled blood flowing in. (Beavers and ducks also make use of this heat-exchange system, in yet another case of convergence.) By these means, leatherbacks can keep their core up to 30 ºC warmer than their skin, evolving some of the same endothermic abilities as characterise mammals.
Turtles are not the only living non-mammals to have shifted from cold-bloodedness (ectothermy) to endothermy (warm-bloodedness). Lamnid sharks also maintain core body temperatures of 20–30 ºC. One such lamnid is the salmon shark, which lives in the cold waters of the north Pacific. It has become so specialised for endothermy that the aerobic, locomotor muscles which power its continuous swimming seem mammal-like (Bernal et al. 2005). The same is true of tunas.
In modern thought, evolutionary change is inextricably linked with the idea that life came into being from primeval chaos. The miracles of evolution are then seen as support for this view. But do natural miracles really have a place in a godless cosmos? Surely the greater the degree of change, the more difficult it is to understand it as unorchestrated. Complex organs such as salt glands, complex systems such as a sea turtle’s temperature control mechanisms, clearly evolved with an end in view, namely to equip a formerly terrestrial animal for life at sea. Atoms have no propensity of atoms to assemble themselves into complex structures, and supposing that they do have that propensity in living organisms merely begs the question. Given that all explanations are after the fact, the more natural explanation is that the innovations were called forth by a pre-existing molecular program. A similar conclusion can be drawn from the development of neural plates but not scutes in the Triassic turtle Odontochelys. Just as plates formed before scutes in the evolutionary history of turtles, so they still do in modern turtle embryos. The evolution of their distinctive carapace was genetically regulated. In not proceeding to the final stage Odontochelys was an example of ‘paedomorphosis’ (Reisz & Head 2008), a common phenomenon in evolution. Some species still retain soft shells.
In Darwinian philosophy new species arise as a result of nature selecting whatever mutation gives the organism in its environment a competitive advantage. Mutations have to build the new organ brick by brick, and species have to wait until completion before profiting by it; there is no support in the fossil record for the concept of half-built organs conferring half the advantage. In the case of land animals sniffing the sea air, there is the additional problem that the sea is a hostile environment: it is cold, its oxygen unusable, its salts poisonous. It requires an ability to swim underwater. There is no incentive to make the business of survival harder by exchanging a tolerant environment for an intolerant one. If the animals making up a species are to be pushed where they will not naturally go, the only way they will master the environment is by acquiring the requisite adaptations quickly and simultaneously.
The record of turtle diversification brings into play questions that have long vexed the palaeontological effort to understand evolution in terms of slow, stepwise change, directed by the vicissitudes of external events. ‘All paleontologists know,’ wrote Stephen Jay Gould, ‘that the fossil record contains precious little in the way of intermediate forms; transitions between major groups are characteristically abrupt’. They also know that science is not, at the highest level of interpretation, about allowing the evidence to speak for itself; it is about making it conform to a preconceived view of life, what Gould termed ‘the essence of Darwinism’. Anything other than a Darwinian explanation would be ‘apostasy’.
Consensus on the evolutionary position of turtles within the amniote phylogeny has eluded evolutionary biologists for more than a century. This phylogenetic problem has remained unsolved partly because turtles have such a unique morphology that only few characters can be used to link them with any other group of amniotes. (Zardoya & Meyer 2001)
The turtle body plan is evidently highly derived, indeed unique among tetrapods. The problem for an evolutionary biologist is to explain these transformations in the context of a gradualistic process. (Rieppel 2001)
Since the fossil record has yet to reveal what kind of animal gave rise to the earliest known turtles, the speculation continues, but whatever the ancestor was, the apparent absence of intermediates between major groups means the transition is likely to have been abrupt.
In recolonisation theory, as in Darwin’s, the history of the lineage giving rise to turtles must go back way beyond the Palaeozoic. No one would wish to argue that evolution started only when the first fossils appeared. Indeed, the placodonts are a parallel case, for while they also do not present a smooth series of intermediates, armoured placodonts do seem to have descended from unarmoured ones. The turtle shell was just one innovation among many. Like the repositioning of the limb girdles, new breathing muscles, two different neck-retraction mechanisms, salt glands, flippers, the trans-oceanic navigation system and leatherback endothermy, the dermal armour of turtles evolved. In evolution anything is possible, even things that seem impossible. The question is: what, or who, was responsible for its wonders.








Comments are closed.