 Mesozoic birds evolved, in a non-incremental way, from theropod dinosaurs. Modern birds are distinct and of unknown origin, though they must have had forbears in the Mesozoic. To the extent that some might look similar to antediluvian birds, that would be through having converged on those original forms after a vast amount of Cenozoic speciation.
Mesozoic birds evolved, in a non-incremental way, from theropod dinosaurs. Modern birds are distinct and of unknown origin, though they must have had forbears in the Mesozoic. To the extent that some might look similar to antediluvian birds, that would be through having converged on those original forms after a vast amount of Cenozoic speciation.Did birds evolve from dinosaurs? Palaeontologists say that they had to have evolved from non-birds, and dinosaurs are the obvious ancestors. For those who believe that animals were created, the question is more difficult. On the one hand, there is an obligation to respect the fossil evidence; on the other, the ancient Hebrew record speaks about an ark on which there were ravens and doves, and since these preceded everything in the fossil record, the conclusion must be that birds as a group could not have evolved from anything – not those birds, at any rate. Or perhaps your reaction is more intuitive. You draw on your knowledge of living birds and think: Are they not miraculous, these gravity-defying wonders of engineering, these masterpieces of colouration, these unwitting musicians from paradise? You marvel at how a sparrow flies into a hedge and processes spatial information faster than lightning as it selects a perch. You recall how swallows every autumn fly south over the English Channel, down western France, across the Pyrenees, through eastern Spain and Morocco, across the Sahara and, if they have not died from starvation or exhaustion, relentlessly on until they arrive in South Africa. How could the frenetic humming-bird, the flamboyant peacock, the woodpecker all have descended from cousins of Tyrannosaurus rex? Having such thoughts myself, I sympathise. Why should we permit reductionism to overrule these other perceptions of reality? If we lived in a world without birds, the very idea of a bird would be unimaginable.
There are around 10,000 species of bird today, and there must have been manifold more in the past, not captured by the fossil record. But what is a bird? Until recently, it seemed sufficient to say, as a kind of short-cut, that a bird was an animal that had feathers, just as all reptiles have scales and mammals hair, the purpose of feathers being of course to enable flight. But life is never so simple. Some reptiles, we have seen, became anatomically mammal-like; in their metabolism some turtles and ichthyosaurs became mammal-like; some fish developed limbs. So feathers might be a defining feature of birds yet not be exclusive to birds. Likewise, birds need more than just feathers to enable them to fly. Among other design features, their extra-strong bones are ‘pneumatised’, crammed with air pockets to reduce the weight of the bones and help with oxygen intake during flight. Crisscrossing struts provide additional strength. Connected with the air-filled bones are four pairs of air sacs and a clavicular air sac, working to maintain a constant unidirectional flow in and out of the lung.
What about feathered animals that cannot fly, such as kiwis? Kiwis are in some ways more like mammals than birds, and their feathers are quite hair-like. In general, there are seven types of feather. Penguins, which are also flightless, have at least four: pennaceous feathers, filoplumes, afterfeathers and plumules (Williams et al. 2015). Pennaceous feathers consist of a symmetrical vane of barbs and interlocking barbules either side of a shaft. In penguins they form a stiff outer layer that protects the skin. Filoplumes, shafts less than a centimetre long, splay terminally into barbs and barbules; they tell the bird when the pennaceous feathers are displaced and need preening, using oil from a gland on the tail. Afterfeathers and plumules are soft downy feathers that insulate the body. All modern bird feathers grow from follicles, including penguin feathers, but none of the penguin feathers function for flight. If a penguin is a bird, it is a very peculiar one, and we do not say that its four different types of feather are evolutionary stages on the way to becoming true flight feathers. Even with birds that fly, feathers can have several other functions, such as waterproofing, thermoregulation, courtship and camouflage.
A flood of new discoveries over the past decades has revealed unexpected complexity. Homoplasy [independent evolution of the same feature] was rampant in early dinosaurian evolution, and the earliest members of the major subgroups were very similar in body size and morphology, which makes unravelling their relationships remarkably difficult. (Baron et al. 2017)
Lagerpetids [a group of dinosauromorphs], pterosaurs, and small- to medium-sized dinosaurs … all lengthened the metatarsus relative to the femur and tibia, in association with increasingly cursorial adaptations. However, it is currently unclear how many times these hindlimb modifications evolved independently, given the complex distribution of character states among these taxa. (Nesbitt et al. 2017)
In the end, the only thing left to signal their origin is the appearance of a hole in the hip socket, articulating the thigh bone. At that stage, we see the almost simultaneous rise of three major groups with a hole in the socket: sauropodomorphs, theropods and ornithischians. While having this in common, the three types of dinosaur differed according to whether their pubis bone projected forwards or backwards. The ancestral  condition was to project down and forwards, as with the dinosauromorphs and as with the sauropods and theropods, termed saurischians. Sauropods were herbivore quadrupeds; theropods, with their sharp teeth and claws, were carnivore bipeds. Ornithischians, on the other hand, were herbivore bipeds and had a pubis projecting backwards, parallel with the ischium. Although analysed as the oldest group, they did not greatly diversify until the start of the Jurassic, by which time they already had a global distribution.
condition was to project down and forwards, as with the dinosauromorphs and as with the sauropods and theropods, termed saurischians. Sauropods were herbivore quadrupeds; theropods, with their sharp teeth and claws, were carnivore bipeds. Ornithischians, on the other hand, were herbivore bipeds and had a pubis projecting backwards, parallel with the ischium. Although analysed as the oldest group, they did not greatly diversify until the start of the Jurassic, by which time they already had a global distribution.
The phylogeny of the earliest dinosaurs remains unresolved. A recent analysis, more comprehensive than previous ones, suggested that there was also a fourth group of dinosaur called herrerasaurids, and sauropods were more closely related to these than to theropods (Baron et al. 2017). This finding came as a shock, so another team was assembled to re-analyse the data. They identified some miscodings and re-affirmed the traditional scheme, but acknowledged that early dinosaur relationships were much less certain than previously supposed; indeed there was even a possibility that sauropods were most closely related to ornithischians (Langer et al. 2017).
We are back to the question of whether dinosaurs really are a single group. When originally proposed, the concept of a common identity was based on six distinctive characters, too many to be regarded as convergent evolution (Bakker & Galton 1974). Now that there is only one diagnostic character, the possibility that the hole in the hip socket might have arisen independently in the earliest sauropodomorphs, theropods and ornithischians, even the herrerasaurids, is not quite so troublesome. Subsequent variation, by contrast, was enormous.
We will assume that modern analyses of the relationships within these lineages are broadly correct, and that there were no more than four kinds of dinosaur. Since the theropods included dinosaurs with feathers as well as others without, there seems little mileage in picking off those that had feathers and arguing that they were separately created.
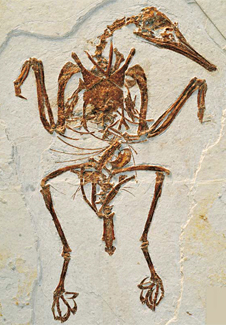
It was in back in 1998 that a team of palaeontologists announced the discovery of ‘feathered dinosaurs’ in China. It stirred a lot of debate. At that time there was still uncertainty as to whether birds evolved from dinosaurs, distinct from some other archosaur, and even those willing to believe that they did were unclear how the new finds fitted into the story. I was lucky enough to see the fossils when they were exhibited in Pittsburgh’s Carnegie Museum of Natural History. A party of us drove over from the creationist conference that was going on just thirty miles from the city.
One of the fossils was Caudipteryx (Ji et al. 1998). It was an odd-looking theropod, about a metre long, with long legs. Primary, symmetrical feathers were attached to the distal part of its relatively short arms and formed a fan at the end of its relatively short tail. Semiplumes and down-like feathers covered the rest of the body. It could not fly, though possibly its ancestors could (so that it had lost the ability to fly). Another of the fossils was Protarchaeopteryx(younger than Archaeopteryx, despite its name). It also bore feathers at the end of its tail and semiplumes and down-like feathers on the rest of the body. Both animals were later classified as early representatives of the oviraptorosaurs. A third, Sinosauropteryx, had ‘integumentary filaments’ (i.e. hair) that were likened to feathers of some sort. Taxonomically, the dinosaur was a compsognathid, a contemporary of Protarchaeopteryx but much further back in the phylogeny. Since 1998, integumentary filaments have been found on several other theropods, including the large Early Cretaceous tyrannosaurid Yutyrannus.
Oviraptorosaurs, named after dinosaurs that were thought to have robbed other dinosaurs’ eggs until further finds showed the eggs were their own, are a group lying evolutionarily within the Maniraptora. The Maniraptora divide into two main branches, one the oviraptorosaurs, the other the paravians (Paraves), a group that gave rise to birds. In addition to feathers, the oviraptorosaurs had other bird-like features, and some were fossilised over nests and eggs.
The most famous ancient bird is Archaeopteryx, from Germany’s Altmühltal Formation, dating to around 150 Ma ago in geological time and discovered just two years after Darwin’s Origin of Species. As in modern birds, its wings had true flight feathers (called remiges, literally ‘rowers’), both primary and secondary, and three claws. The long tail, too, had flight feathers (retrices, literally ‘helmsmen’, helping to brake and steer); some of these were symmetrical, some asymmetric. In common with most birds and certain other theropods it had a furcula or wishbone and low bone density. The most puzzling thing about the fossils from China was that they dated to the Lower Cretaceous, some 25 Ma after Archaeopteryx in the Late Jurassic. Many of the experts (not all) found it difficult to regard Caupdipteryx and the prejudicially named Protarchaeopteryx as intermediates on the way to becoming birds when Archaeopteryx was already capable of flight.
 Debate as to how well Archaeopteryx and other bird-like dinosaurs from the Mesozoic could fly simmers on, and new, ever more comprehensive analyses go on revising the relationships. However, the hope has long gone that these would reveal a single stepwise progression towards powered flight, and bigger analyses are not necessarily better. This is because ‘large-scale analyses of equally weighted morphological characters run the risk that many simple homoplastic characters overrate fewer ones of greater phylogenetic significance’ (Mayr 2009). One analysis divides the paravians into deinonychosaurs and avialans, groups which diverged around the Middle Jurassic (Hartman et al. 2019). Among the deinonychosaurs, only Archaeopteryx, the Early Cretaceous Microraptor and the Late Cretaceous Rahonavis are judged to have been capable of powered flight, whereas, as the name implies, nearly all avialans flew. The three deinonychosaurs each acquired the ability independently, and all were dead-ends: they were not the progenitors of later species that flew. Oviraptorosaurs could have been ancestral to the paravians, but do not appear as fossils until the Cretaceous. They may have lost the ability to fly, in which case the origin of their volant ancestors is as obscure as that of the oldest known bird.
Debate as to how well Archaeopteryx and other bird-like dinosaurs from the Mesozoic could fly simmers on, and new, ever more comprehensive analyses go on revising the relationships. However, the hope has long gone that these would reveal a single stepwise progression towards powered flight, and bigger analyses are not necessarily better. This is because ‘large-scale analyses of equally weighted morphological characters run the risk that many simple homoplastic characters overrate fewer ones of greater phylogenetic significance’ (Mayr 2009). One analysis divides the paravians into deinonychosaurs and avialans, groups which diverged around the Middle Jurassic (Hartman et al. 2019). Among the deinonychosaurs, only Archaeopteryx, the Early Cretaceous Microraptor and the Late Cretaceous Rahonavis are judged to have been capable of powered flight, whereas, as the name implies, nearly all avialans flew. The three deinonychosaurs each acquired the ability independently, and all were dead-ends: they were not the progenitors of later species that flew. Oviraptorosaurs could have been ancestral to the paravians, but do not appear as fossils until the Cretaceous. They may have lost the ability to fly, in which case the origin of their volant ancestors is as obscure as that of the oldest known bird.
In addition to their occurrence in the four species mentioned, asymmetric feathers, usually a good indication of powered flight, also evolved independently in Jianianhualong (a troödontid) and in avialans such as Archaeornithura and Chongmingia. That too is surprising, for the appearance of asymmetric feathers was ‘one of the major events in feather evolution’ (Xu et al. 2017).
The complex structure of avian feathers, including all information from fossilized feathers such as those present in Archaeopteryx, suggests strongly that feathers evolved only once in the history of the Vertebrata. This conclusion is based on the complicated morphology present in all feathers, as well as their particular and intricate mode of embryological development. (Bock 2000)
The small flight feathers on the alula, which give added control at low speeds, also evolved in Microraptor and some avialans independently. Convergences bedevil evolutionary schemes however the data are ordered. Nor should one forget that feathers are attached to muscles and muscles to bones, all of them specific to flight, and the whole apparatus controlled by the brain. The evolution of birds entailed the coordinated transformation of every part of the anatomy. As with the evolution of dinosaurs as a whole, every new find ‘demonstrates that the early evolution of birds was complex and homoplastic’.
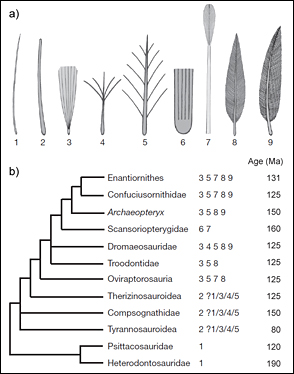 A common term in the literature is ‘mosaic evolution’. The idea is that a body develops as modules, and that these evolve more or less independently. The cranium of a bird, for example, consists of seven such modules, and depending on the species has evolved at different rates. More loosely, when a fossil is described as a mosaic, the reference is to a novel combination of features that themselves are not all novel. As with convergence, the frequency of the phenomenon frustrates the search for a linear story. Despite its celebrity status, Archaeopteryx is hard to classify because few of its characters are unique in themselves; it is only the particular combination of characters that make it unique (Rauhut et al. 2018). Some workers therefore analyse Archaeopteryx as a deinonychosaur (as in the Hartman cladogram), others, as an avialan (e.g. Wang et al. 2016). Wherever it is placed, it remains a surprisingly early example of powered flight, and fossils do not show a progressive evolution of feathers leading up to it, though the hind limbs of Serikornis and both the front and hind limbs of Anchiornis, animals apparently related to Archaeopteryx, bore pennaceous feathers and may have been capable of gliding. Yutyrannus had filaments of some kind but was not an ancestor of Archaeopteryx, and later tyrannosaurids such as Tyrannosaurus had scales. Thus the whole notion that simple filaments evolved step by step to complex feathers is baseless. Assertions of progressive evolution towards barbed vanes are disingenuous (e.g. Xu et al. 2010), and fail to recognise that each of the different structures must have had a purpose of its own, a purpose that could have had nothing to do with functions that still lay in the future. Each species was a viable organism in its own right. We should not be imagining animals comically leaping and flapping their arms, or launching themselves off branches – like early attempts at human-powered flight – in an effort to become something they were not.
A common term in the literature is ‘mosaic evolution’. The idea is that a body develops as modules, and that these evolve more or less independently. The cranium of a bird, for example, consists of seven such modules, and depending on the species has evolved at different rates. More loosely, when a fossil is described as a mosaic, the reference is to a novel combination of features that themselves are not all novel. As with convergence, the frequency of the phenomenon frustrates the search for a linear story. Despite its celebrity status, Archaeopteryx is hard to classify because few of its characters are unique in themselves; it is only the particular combination of characters that make it unique (Rauhut et al. 2018). Some workers therefore analyse Archaeopteryx as a deinonychosaur (as in the Hartman cladogram), others, as an avialan (e.g. Wang et al. 2016). Wherever it is placed, it remains a surprisingly early example of powered flight, and fossils do not show a progressive evolution of feathers leading up to it, though the hind limbs of Serikornis and both the front and hind limbs of Anchiornis, animals apparently related to Archaeopteryx, bore pennaceous feathers and may have been capable of gliding. Yutyrannus had filaments of some kind but was not an ancestor of Archaeopteryx, and later tyrannosaurids such as Tyrannosaurus had scales. Thus the whole notion that simple filaments evolved step by step to complex feathers is baseless. Assertions of progressive evolution towards barbed vanes are disingenuous (e.g. Xu et al. 2010), and fail to recognise that each of the different structures must have had a purpose of its own, a purpose that could have had nothing to do with functions that still lay in the future. Each species was a viable organism in its own right. We should not be imagining animals comically leaping and flapping their arms, or launching themselves off branches – like early attempts at human-powered flight – in an effort to become something they were not.
But while Darwinian evolution cannot have an end in view, pre-programmed evolution can. From a Darwinian perspective the appearance of pennaceous feathers in some lineages before powered flight, even in lineages that never gave rise to powered flight (compsognathids, oviraptorosaurs, troödontids), is puzzling, for how could natural selection have led to a design so specifically geared to flight when the animal itself could not fly? That is why some feathered dinosaurs are sometimes interpreted to have lost the power of flight. If, on the other hand, flight was always in mind for some species, the appearance of feathers serving a more general purpose in advance of flight is comprehensible. Instructions for scales, hair and feathers were in the genome of all theropods from the beginning, ready to be activated at any time in concert with whatever made up the species.
Given that pelvis design determines the primary division of dinosaurs into saurischians and ornithischians, it it may seem odd that birds evolved from the saurischians, which had reptilian hips, rather than from ornithischians, which had beaks and (as their name signifies) bird-like hips. The solution of this anomaly is that some theropods acquired bird-like hips convergently with ornithischians before evolving into birds. Indeed, they did so no fewer than three times: within alvarezsauroids, within dromaeosaurids, and within troödontids (just once, in Sinovenator). In each of these cases the basal members of the group had the normal orientation. The downward-pointing pubis of Archaeopteryx was intermediate (Macaluso & Tschopp 2018), while Cretaceous birds (e.g. Confuciusornis) represent a fourth instance. To confuse matters further, although ornithischians had nothing to do with bird evolution, some were covered in ‘filamentous integumentary structures’ (Zheng et al. 2009).
The backward orientation of the pubis had two main advantages for birds. One is that it increased the efficiency of the tail muscles, enhancing their ability to take off from the ground; the other is that it enabled the pelvic girdle during flight to participate in driving air into and out of the lungs. Flying is energy-intensive and requires a lot of oxygen.
The avialans of the Cretaceous comprise (1) the family that includes Confuciusornis, (2) enantiornithines, and (3) ornithuromorphs. The co-existence of three distinct bird groups was not predicted when, in the 1970s, palaeontologists first concluded that birds evolved from dinosaurs. All three first appeared in the Early Cretaceous and were restricted to the Cretaceous. Confuciusornithids are known only from the Early Cretaceous and are not discussed further.
Modern palaeontology rests on the assumption that all species are related, be it distantly or closely, so the a priori commitment to unlimited evolution limits interpretation. That birds might have had more than one origin is not considered. Nonetheless suppose, by way of a thought experiment, that all things owed their existence to a supernatural being, and that that being might have wished to demonstrate that he was not limited either in imagination or power, that he could as easily evolve birds from some other group as speak them instantaneously into being. After all, if creation ex nihilo is difficult, how much more difficult would it be to devise a biological program capable of causing the things that make for a bird to evolve: the requisite feathers, muscles, skeleton, metabolism, lungs, blood vessels, oesophageal crop, double-chambered stomach, lightning-quick brain. There would then, as regards ultimate ancestry, be at least two groups of bird, one created and one evolved. Necessarily, having converged on very similar anatomy, they would be difficult to distinguish, but they would be distinguishable, because that, in part, was the intention: to make clear the answer to his own question, “Is anything too hard for me, the God of all flesh?”. Although there might not be great discontinuity between dinosaurian and non-dinosaurian birds, they would differ clearly enough for their independent origins to be apparent. Ideally, the discontinuity would be chronological as well as morphological.
The cumbersome name enantiornithes, meaning ‘opposite bird’, refers to the way the shoulder bone articulates with the strut-like coracoid that links it with the sternum/breastbone. (Click on the image at the head of this article.) The sternum provides an attachment surface for the wing muscles. In enantiornithes the articulating end of the coracoid is convex and the corresponding part of the shoulder-bone concave, whereas in modern birds the joint is the other way round. Like Archaeopteryx, most species had teeth and clawed fingers on the wings, and specialisations for perching suggest that they lived among trees. Flight-enhancing features in the enantiornithines such as a deeply keeled sternum (absent from Archaeopteryx), narrow furcula, and quill knobs on the ulna are also seen in modern birds, where they are regarded as convergent (Atterholt 2018), but the pattern of sternum ossification was unique. The origins of the group are obscure. Like both Archaeopteryx and confuciusornithids, enantiornithines are not preceded by a progressive series of steps towards flight. Despite their flying ability, early diversification and worldwide distribution, they became extinct at the end of the Cretaceous. They are not the ancestors of modern birds.
Since the enantiornithines went extinct, discontinuity between them and modern birds is obvious enough. The critical group is therefore the ornithuromorphs. The oldest known example is the long-legged wading bird Archaeornithura, dated to 131 Ma, some of whose features were surprisingly advanced. Four distinctive features unite the group, of which all but one appear, separately, in other taxa (O’Connor & Zhou 2013). That there is not a lot of difference between the enantiornithines and ornithuromorphs is illustrated by Cruralispennia. Although an enantiornithine, the bird had a plough-shaped pygostyle (a bone consisting of fused vertebrae that support the tail feathers) characteristic of ornithuromorphs, ‘highlighting the homoplastic nature of early avian evolution’ (Wang et al. 2017). In the Hartman analysis the two groups are lumped together under the label ‘Ornithothoraces’.
Various sites from western North America indicate that archaic ornithuromorphs persisted to the end of the Cretaceous. Sparse and fragmentary though the record is, species numbers do not appear to have been in decline (Longrich et al. 2011). Attempts to assign the fragments to modern bird groups have been unsuccessful. One reflection of the discontinuity is the conflict between ‘molecular clock’ predictions of when modern birds should appear (having diverged from ornithuromorphs) and when they actually appear: 100 Ma ago per the molecules (Feduccia 2003, Jarvis et al. 2014), around 60 Ma say the fossils. This is after imposing a ‘maximum age constraint’ on the clock; without the constraint, the molecular evidence would put their appearance around 160 Ma ago (Cracraft et al. 2015). In other words, the first modern birds represent a difference equivalent to 100 Ma of evolution.
- The ankle and foot bones are fused.
- Flight feathers are asymmetric and, while these are similar to the asymmetric feathers of Mesozoic birds, the barb angles of the trailing vanes tend to be significantly larger, enhancing wing aerodynamics (Feo et al. 2015).
- Mesozoic birds were heavier, possibly too heavy to incubate their eggs (Deeming & Mayr 2018).
- Most Mesozoic birds had teeth (not the Confuciusornis family). All extant neornithines are toothless.
- The neornithine beak is mostly an elongation of the dual, usually fused, premaxillary upper jaw bones rather than of the maxilla behind them. Hinging with the cranium allowed the beak to be moved separately.

Most of the other neornithine orders are attested from the early Eocene (Mayr 2014). Only one Cretaceous fossil is unequivocally identifiable as a modern bird (Field et al. 2020). Consisting of a nearly complete skull, it shows a previously unseen combination of features diagnostic of both galliforms (pheasants and their kin) and anseriforms (ducks, geese and swans), thereby confirming the hypothesis that the two groups are related. However, it does not narrow the gap between ornithuromorphs and modern birds, since almost none of the cranial anatomy falls outside the range of extant galloanserans. It is dated to 66.7 Ma, shortly before the extinctions at the end of the Cretaceous. A fragmentary fossil of the same age includes a toothed beak but neognath-like palate and does suggest a link between ornithuromorphs and galloanserans (Benito et al. 2022). “This fossil shows that the mobile beak – a condition we had always thought post-dated the origin of modern birds, actually evolved before modern birds existed.”
Neornithines form two major groups, Palaeognathae and Neognathae. Palaeognaths, or ratites, comprise ostriches (Africa), tinamous (South America, up to Mexico), rheas (South America), emus and cassowaries (Australia, New Guinea), and kiwis (New Zealand). As with humans, their palate bones are fused together. Neognath palate bones are connected by a mobile joint. According to the molecular evidence, ratites, which were once proficient flyers, became irrevocably flightless along at least three separate lineages, the extinct moa going so far as to lose its wings completely. Gigantism, an advantage to birds unable to fly from danger, also developed independently. Their genetic programs pushed many of the ratites into mimicking ornithomimid theropods, as if evolution were going backwards: hence, looking forward, the ornithomimid Rativates, of the Late Cretaceous, means ‘prophetic of the ratites’; its contemporary Struthiomimus, ‘ostrich-mimic’. Ostriches were probably the first to attain their distinctive form, kiwis the last. While they encompass a great range of forms and sizes, monophyly of the palaeognaths is well supported.
Neognathae comprise all other birds, differentiated primarily in respect of their jaws. Morphological differences within the group enable up to thirty orders to be distinguished, for example loons, grebes, penguins, parrots, cuckoos. How the orders relate to one another is controversial. A ‘pessimistic’ summary of the state of knowledge in 2004 described them as forming a comb rather than a tree; it was not possible to distinguish a unifying phylogeny (Cracraft et al. 2004). That remains the position. Morphological convergence is ‘rampant’, not least because the molecular evidence – thought to be more dependable than morphology – splits similar-looking species of the same order, such as hawks and falcons, into widely separated orders. While molecular analyses appear to have resolved relationships between lower-level taxa, higher-level relationships remain elusive, frustrated by ‘massive protein-coding sequence convergence’ (Jarvis et al. 2014, Han et al. 2011). The problem of convergence, in other words, does not go away with this type of analysis. Molecular studies do not even agree with each other. Large-scale analyses being costly, some teams restrict themselves to obtaining many molecular characters from a small number of representative species, others to obtaining few characters from a large number of species. Also important is whether mainly non-coding or mainly coding parts of the genome are analysed (Reddy et al. 2017).
This is not to say that all the orders are unrelated. But where relationships are proposed, molecular and morphological evidence broadly need to agree, for genes express morphology, and natural selection, in principle, works on the phenotype, not the genotype. Analysis has failed to produce a unified tree because the genomes chosen as representative of the orders – complex 3-dimensional networks, not mere strings of information – are uniquely organised; they were not the product of gradually accumulating codon substitutions, each mutating in parallel. The irreducibility of the orders to a single tree suggests that the whole exercise of translating molecular differences into divergence times may be misconceived.
Unique organisation is what one would expect if a deity had created several distinct kinds. Otherwise it would have been more parsimonious to have created just one kind and provided for every order to have evolved from that. Here too the data are trying to tell us something. Indeed, one order, Passeriformes, seems to have been created specifically to demonstrate that God could have brought all species out of just one kind, had he wished. All by itself this order comprises 137 families and accounts for more than half of all living neornithine species. Passerines began diversifying, it is thought, in Australia, in the early to mid Eocene, and to have split soon thereafter into three suborders: New Zealand wrens (now just 2 species), oscines (5000 species) and suboscines (1250 species). Distinct families began to differentiate in the late Oligocene (Oliveros et al. 2019). Oscines are characterised by their distinctive vocal organs and complex songs, often learned through imitation. Among the least musical are the crow family, many of them rather drab animals but closely related to the birds of paradise. Suboscines have less complex vocal organs and apparently do not learn their songs by imitation. They consist of two groups, the New World suboscines, mostly restricted to the tropical forests of Central and South America, and the Old World suboscines, chiefly inhabitants of eastern Africa and southeast Asia but formerly also Europe.
The same message of infinite power is conveyed by other species-rich groups in the animal kingdom: beetles among insects (1,500,000 species, 25% of the total), rodents among modern mammals (almost 2,300 species, 40% of the total) and teleosts among fishes (26,000 species, 95% of the total). This is just counting living species. Conversely, exceptionally low-diversity groups demonstrate that there is nothing inevitable about evolution; chance mutations are not continually throwing up new material on which natural selection can act. One bird order, the hoatzins, consists of just one living species. Its relationship within the putative neornithine tree is unresolved (Prum et al. 2015).
Ravens are part of the crow family, Corvidae, hence highly evolved passerines whose ancestors would not have looked anything like ravens. Those on Noah’s ark therefore must have died out, and modern ravens subsequently converged on the same form. Doves are part of the Columbidae family, consisting of doves, pigeons and the flightless dodo. The order Columbidae consists solely of this family, though it is probably related to sandgrouse and mesites, forming the superorder Columbimorphae. In molecular analyses it appears to be one of the oldest orders, about the same age as galloanserans. Some species, such as turtle doves and pigeons, find their way over long distances by orienting themselves to the Earth’s magnetic field, an ability convergent with that of Hirundinidae (swallows and martins) and many other migratory birds, not to mention whales, sea turtles, bees. Ravens and doves are evidence that some antediluvian lineages survived up to the present day with little change, while others diversified enormously. Assuming that the Hebrew word for raven denotes the same form then and now, God had the power to create by evolution the very same forms he created by fiat.
The explosive radiation of so many orders after the K/T boundary, apparently out of nowhere, is not an isolated phenomenon. Similarly explosive was the appearance of marine invertebrates in the Cambrian, of vascular plants, insects, amphibians and amniote reptiles in the Devonian-Carboniferous, of dinosaurs in the late Triassic and early Jurassic, of flowering plants in the mid Cretaceous, of modern mammals in the early Cenozoic. The parallel with the mammals is particularly relevant, because, as with enantiornithine and ornithuromorph birds, mammals in general are attested before the new group appear. Just as Mesozoic mammals directly preceded modern mammals but were not ancestral to them, so Mesozoic birds were not ancestral to modern birds.
No one knows for sure why the neornithine record does not extend back into the Mesozoic, nor why the catastrophe at the end of the Cretaceous killed off archaic mammals, pterosaurs, dinosaurs and their winged relatives but not all modern mammals and neornithines. But three factors can be suggested: metabolism, geography and diet.
First, metabolism. Like modern mammals, birds are warm-blooded animals. In scientific terminology they are ‘endotherms’ (heated from within), whereas reptiles are cold-blooded animals or ‘ectotherms’ (heated from outside). But we have already seen that the dichotomy is not absolute: some animals, for example turtles and lamnid sharks, dubbed ‘mesotherms’ have the capacity to raise their body temperature for a time but not keep it constantly high. Dinosaurs, the evidence suggests, were of the latter type.
Metabolic rate scales with growth rate, and when the variables are plotted against each other, living animals are distinct: endotherms grow fast and have high metabolic rates, ectotherms grow slowly and have low rates. A few species fall inbetween. In the case of the dinosaurs, growth rate can be determined from bone size and from the annual rings in the bones. It turns out that they too are intermediate, although large dinosaurs, such as sauropods, probably had higher metabolic rates than small dinosaurs (Grady et al. 2014). That the dominant terrestrial animals were mesothermic makes sense in a world where the temperature gradient from equator to pole was much lower than today, and where the climate generally was warm. Dinosaurs even inhabited Alaska and Antarctica. They did not need to generate a lot of heat themselves, and many of them had feathers for insulation. Neornithines, on the other hand, with their high metabolic rates, might not have been comfortable in the lower latitudes of a greenhouse world.
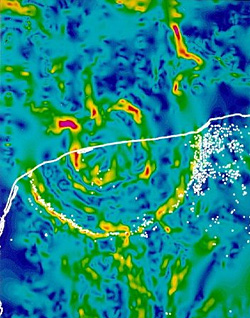 By the same token, a sudden temperature downturn might have been catastrophic for mesotherms. The cause of the end-Cretaceous extinctions was an asteroid that gouged out a 190-km crater off the coast of Mexico. Falling ejecta heated the atmosphere and kindled wild fires, after which came an ‘impact winter’, as dust and aerosols clouded the stratosphere and blocked the Sun’s radiation (Vellekoop et al. 2014). Deprived of solar energy, great tracts of vegetation were laid waste. Terrestrial animals hardest hit by the cold would have been the mesotherms, unable to generate enough of their own heat as modern birds and mammals could, and unable to hibernate through it as snakes and lizards could. Large animals would have starved to death. The metabolism of the enantiornithines and ornithuromorphs was probably somewhere between that of dinosaurs and modern birds; they too were vulnerable.
By the same token, a sudden temperature downturn might have been catastrophic for mesotherms. The cause of the end-Cretaceous extinctions was an asteroid that gouged out a 190-km crater off the coast of Mexico. Falling ejecta heated the atmosphere and kindled wild fires, after which came an ‘impact winter’, as dust and aerosols clouded the stratosphere and blocked the Sun’s radiation (Vellekoop et al. 2014). Deprived of solar energy, great tracts of vegetation were laid waste. Terrestrial animals hardest hit by the cold would have been the mesotherms, unable to generate enough of their own heat as modern birds and mammals could, and unable to hibernate through it as snakes and lizards could. Large animals would have starved to death. The metabolism of the enantiornithines and ornithuromorphs was probably somewhere between that of dinosaurs and modern birds; they too were vulnerable.
Second, geography. In general, populations will only speciate if they become isolated from one another and cease to interbreed. The fact that neornithines did not noticeably impinge on the fossil record in the Mesozoic suggests that they were few in number and not geographically widespread. Their rapid diversification early in the Cenozoic suggests that they then spread and multiplied far and wide.
It must also be remembered that we know so much about dinosaurs because fossils of them are abundant. That is partly because they were themselves abundant and partly because they lived in environments which, from time to time, were prone to floods and sandstorms, or lived in environments which were inherently more conducive to fossilisation, such as oxygen-poor lagoons; they will not have lived in every type of environment. It could be that most modern birds and mammals lived in continental interiors where the chances of fossilisation were poor. Elevated areas, for example, are places where fossilisation hardly ever occurs. Bird fossils from the first half of the Cenozoic come mostly from lacustrine or marginal-marine sediments, suggesting that in greater numbers they were now spreading into habitats previously dominated by pterosaurs. Although the bird fossil record is generally meagre, one early-Eocene coastal site in Denmark yielded the remains of 11 neornithine orders (Dyke & Lindow 2009).
The different orders need not all have spread out from the same area in the Cenozoic, though they must have done originally. Probably the first to speciate in any numbers were the palaeognaths. While biogeographical inferences must be tentative, the majority of fossils occur in the southern hemisphere, including the oldest. A few fossils of Eocene age occur in the northern hemisphere. Dispersal from their geographical origin must have begun before the Cenozoic, since Australia, New Zealand and South America had begun to separate already in the Late Jurassic (Müller et al. 2019). It is difficult to argue that the palaeognaths crossed large expanses of water to reach these landmasses, especially if they had already lost, or begun to lose, the power of flight. The earliest fossils of the extinct ‘terror birds’ come from Africa, South America and Europe, dating to the Eocene. They are presumed to have originated in Africa and, since they were flightless, to have rafted across the Atlantic on floating vegetation (Angst et al. 2013). Passerines, we saw, appear to have originated in Australia, but to have diversified on a large scale only from the late Oligocene. The hoatzin record consists of just three fossils, the oldest coming from Brazil and dating to the transition from Oligocene to Miocene, the next oldest from Namibia. These birds could not fly long-distance, so again the standard explanation is that one or the other species rafted across the Atlantic, choosing not to fly off as the raft drifted seaward. This implausible scenario is invoked only because of the assumption that neornithines had a common origin going back no further than the Late Cretaceous. Without that commitment, the evidence would suggest that hoatzins – just like palaeognaths and terror birds – were already in existence when South America and Africa were one continent, in the Early Cretaceous, even though we have no fossils from that time.
Climate was warm in the Palaeocene because of rapid rates of seafloor spreading, which heated the ocean from the bottom up and kept it much warmer than today. Global temperatures surged at the end of the Palaeocene, when massive volcanism in the North Atlantic caused CO2 to reach 1400 ppm. Thereafter CO2 levels subsided, rose again towards the end of the Eocene, plunged at the Eocene/Oligocene boundary, then fell more gradually towards modern levels. As seafloor spreading slowed, oceans cooled, and temperature differences between low and high latitudes increased. Birds were designed to diversify in such a way as to take maximum advantage of the increasing diversity of habitable environments, and they did so primarily from the Oligocene.
The third factor helping to explain the radiation of the neornithines is the rise of the angiosperms. Angiosperm seeds grow in an ovary embedded in a flower. After fertilisation, the other parts of the flower fall away and the ovary swells to become a fruit. In the Early Cretaceous most angiosperm seeds were small; during the Late Cretaceous to early Eocene (80–55 Ma) the average size of seed increased one hundred-fold (Eriksson 2016). This was also the time when angiosperm-dominated forests reached their maximum extent.
Not all angiosperms produce fleshy fruit, not all birds are frugivores, and neornithines, especially neognaths, were not the first birds to have fed on seeds and fruit. Nonetheless, neornithines as a class benefited greatly from these developments, just as angiosperms benefited greatly from the dispersal of their seeds by the birds. A large part of bird diversity today is concentrated in the tropical rainforests of South America, Africa, southeast Asia and Australasia. Whether the forests date back further than the Cenozoic is unknown, but neognaths evolved every possible permutation of beak in order to exploit the new vegetation, just as insects in the Cretaceous evolved a great variety of mouthparts for feeding on the nectar.
There is also the issue of time. If the Earth is billions of years old, common descent is the inevitable corollary; if it is much less old, we can free the mind to entertain other possibilities. I have already introduced the thought that time as measured by the radioisotope clock might have been decelerating. We see further evidence of this in the way so much evolution seems to be packed into the later part of Earth history, as witness the diversification  of passerines over the ‘25 million years’ from the late Oligocene onwards. When trying to make sense of the late appearance of any major group in the record, we should bear in mind the possibility that time in the earlier part is much more compressed. Evolution may appear to have been speeding up because geological time was slowing down.
of passerines over the ‘25 million years’ from the late Oligocene onwards. When trying to make sense of the late appearance of any major group in the record, we should bear in mind the possibility that time in the earlier part is much more compressed. Evolution may appear to have been speeding up because geological time was slowing down.
Across the entire fossil record the story is much the same. There were of course other factors at work, including spasmodic mass extinctions in the wake of which fresh waves of diversifying animals took the place of the dead. A chart showing the number of terrestrial and marine families through the last 600 million years of geological time will not be a simple curve, but reflect initial colonisation of vacant ecospace and subsequent periods when organisms diversified prolifically but were repeatedly set back. From the mid Jurassic (170 Ma) originations increasingly outnumbered extinctions, both on land and in the ocean – squamates, crocodilians, dinosaurs, flowering plants, pollinating insects, leaf-eating flies, lepidopterans, beetles, teleosts, mammals, birds. In the terrestrial realm the number of families captured by the fossil record increased sevenfold. Is the pattern of exponential growth real, or an artefact of the way time is measured? The first 3300 million years of geological time are virtually empty. If natural selection working upon mutant genes can do anything given enough time, that is a very long time to be doing virtually nothing.
Did birds evolve from dinosaurs? We need to specify, which birds. If we assume that birds are a single group with a single ancestor, the answer is more or less predetermined, the record being what it is. But birds do not present themselves as a single group, least of all in the Cenozoic. To say that ‘Birds evolved from dinosaurs, and therefore are dinosaurs’ is unthinkingly to lump Mesozoic and Cenozoic birds together and confuse ancestry with identity. Even if the statement were true, it would be like saying that human beings are bacteria because they evolved from bacteria.
Modern birds have no obvious origin. There is some evidence that archaeopterygids, confuciusorniforms, enantiornithines and ornithuromorphs evolved from dinosaurs, even though they are not preceded by a series of intermediates, but these were all dead-ends: they did not give rise to modern birds. Cretaceous ornithuromorphs were the most similar to modern birds, but so different from them that nearly every specialist believes that the two groups co-existed. An ancestral relationship between ornithuromorphs and modern birds has not been established.
Meanwhile, there is again the problem of convergence: features appearing out of nowhere as if they were always latent, then disappearing only to appear in another part of the tree. How could two or more different species take different genetic paths, yet arrive by chance at a complex set of instructions that re-invent the same structure – for example, feathers complete with barbules and interlocking hooklets? Convergence is pervasive in evolutionary phylogenies and undermines the idea that evolution is a Monte Carlo game. Consequently, it is hardly out of line to suggest that neornithines, reducible perhaps to 20-24 orders, are simply another example: 20-24 independent origins of the bird form, adding to the four theropod lineages leading separately to Archaeopteryx, Rahonavis, Microraptor and avialans.
Dinosaurs were not like human beings who raised their eyes to heaven and thought: “Space is a hostile environment, but we will come up with the technology to conquer it, however difficult the task.” They were not engineers of their own biology, intent on expanding their horizons. There was no ‘selection pressure’ forcing dinosaurs to come up with ‘solutions’ – the metaphors typically used to generate the illusion of an explanation. They conquered the air because their Creator enabled them to do so.
Nor were they the only animals to perform the miracle. Insects did it in the Carboniferous, if there were any that started off without wings and acquired them later. Pterosaurs did it in the Middle Triassic, probably evolving from lagerpetids (Ezcurra et al. 2020). Bats did it in the Eocene. In these cases, the fossil record gives us no clues as to how it happened, whether all at once or by small steps too rapid to be captured. When the first winged insects, the first pterosaurs and the first bats made their existence known, they had already performed the miracle. In this respect the theropods are a partial exception. While they do not present us with a chronologically consistent progression from earth-boundedness to powered flight, they do at least prove that the genetics driving their transformation pre-existed.







