
Ichthyosaurs – the name means ‘fish lizards’ – were reptile inhabitants of the sea, at the same time as pterosaurs glided in the air and dinosaurs walked on the land. They looked rather like modern dolphins. The smallest was around 1 metre, the biggest, Ichthyotitan, up to 25 metres, both extremes arising early in their evolution. Ichthyosaurs had eyes as big as those of any animal known, the record being held by the 9-metre-long Temnodontosaurus, whose eyes measured 26 cm across. As it happens, Temnodontosaurus was the very first ichthyosaur to be discovered, by 12-year-old Mary Anning in 1811, who found remains of its skull and vertebrae on England’s Dorset coast.
Despite being well-adapted to the sea, ichthyosaurs must once have been land-lubbers. They had two pairs of limbs, with digit-like bones rather than rays in their flippers. Initially the bones comprised five rows, as in the reptile hand, later up to ten rows. A shoulder girdle connected the body to the skull, the roof of which had a pair of small fenestrae typical of reptiles. Since most reptiles are diapsids, ichthyosaurs presumably lost the lower pair. The small conical teeth were also reptilian.
Another indication of their terrestrial origin is their lack of gills. Like marine mammals, they had to draw oxygen from the atmosphere. When a baby ichthyosaur uncurled itself in preparation for birth, it instinctively oriented itself so that it passed out of the womb tail first. Only as its head emerged did it need to breathe, at which point it immediately swam to the surface, its body parallel with the mother’s.
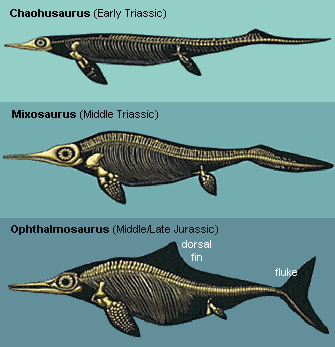 Amongst the oldest known ichthyosaurs was Utatsusaurus, from the Lower Triassic of Japan. As in terrestrial reptiles, the pelvic girdle was attached to the spine, but no longer robustly enough to support the body’s weight. Another transitional feature was the equal length of humerus and femur. In most terrestrial animals the femur is longer than the humerus and the hind limb as a whole larger than the front limb, whereas in the more advanced ichthyosaurs, including the contemporary Chaohusaurus, the humerus was longer than the femur. Utatsusaurus was also closer to terrestrial animals in not having a dorsal fin or a tail fluke. On several fronts, the view that ichthyosaurs were former land-dwellers that evolved adaptations for life at sea is therefore well supported.
Amongst the oldest known ichthyosaurs was Utatsusaurus, from the Lower Triassic of Japan. As in terrestrial reptiles, the pelvic girdle was attached to the spine, but no longer robustly enough to support the body’s weight. Another transitional feature was the equal length of humerus and femur. In most terrestrial animals the femur is longer than the humerus and the hind limb as a whole larger than the front limb, whereas in the more advanced ichthyosaurs, including the contemporary Chaohusaurus, the humerus was longer than the femur. Utatsusaurus was also closer to terrestrial animals in not having a dorsal fin or a tail fluke. On several fronts, the view that ichthyosaurs were former land-dwellers that evolved adaptations for life at sea is therefore well supported.
A number of specimens are preserved with embryos. One pregnant Chaohusaurus – found north of the city of Chaohu, China – made the news because the trove included no fewer than 80 skeletons. In this case the embryos left the womb head-first, as with most land animals, indicating that a tail-first exit was a later development. As with the large assemblage of Shonisaurus fossils in Berlin-Ichthyosaur State Park, Nevada (Kelley et al. 2023), the site may have been a breeding ground.
The earliest fossil ichthyosaurs were small (around 1 m long) and relatively slender. Comparisons with similarly shaped fish, such as catsharks, suggest that they swam by wiggling their bodies sideways, like modern-day lizards. As they chased their prey, their small vertebrae gave them the flexibility to accelerate or change direction quickly. But they did not stop at the shallows. By the end of the Triassic ichthyosaurs were venturing far beyond the continental shelf and, despite having to surface for air, penetrating the ocean down to hundreds of metres. Some ichthyosaurs were as long as today’s blue whales. In the space of 50 million years these terrestrial lizards had become masters of the pelagic environment the equal of any animal.
Because undulatory swimming creates more drag and consumes more energy, it is a poor choice for predators in the open ocean, where food is less abundant. Predators such as the great white shark, a member of the lamnid family, therefore have a stiffer construction: their tails beat sideways but the rest of the body moves little. Their bodies are thicker, their vertebrae larger and stubbier. Together with their crescent-shaped tail fins, these differences help them, as they search for prey, to cruise over large distances with maximum efficiency.
Remarkably, although unrelated to sharks, ichthyosaurs acquired the same adaptations over time: thicker bodies, larger vertebrae, a dorsal fin and a caudal fluke. By increasing both heat conservation and oxygen storage, their larger mass enabled them to plunge to greater depths. Oceans become cooler with depth, and reptiles are cold-blooded: they do not have heat engines and temperature regulation systems to keep their body at a constant temperature. Consequently the first ichthyosaur reptiles were ill-suited to exploiting deep water. Later species put on blubber and evolved an ability to keep their bodies warm. Although greater volcanism on the seafloor meant that oceans were shallower and warmer than today, much below the surface layer they would still have been chilly. It has been calculated that a 4-metre Ophthalmosaurus, one of the most advanced ichthyosaurs, could stay submerged for at least 20 minutes,  time enough to dive down to 600 metres and back. Their huge eyes were highly light-sensitive and capable of resolving fine detail, ideally suited to focusing on small, quick prey in the dimmest regions. As well as additional sensor cells around the nose, an elaborate system of what may have been electro-receptors similar to the systems certain sharks have for detecting electric fields compensated for their inability to see directly in front. The fully evolved caudal fluke was very similar in shape to that of lamnid sharks but differed in that the vertebral column passing through it developed a downward kink.
time enough to dive down to 600 metres and back. Their huge eyes were highly light-sensitive and capable of resolving fine detail, ideally suited to focusing on small, quick prey in the dimmest regions. As well as additional sensor cells around the nose, an elaborate system of what may have been electro-receptors similar to the systems certain sharks have for detecting electric fields compensated for their inability to see directly in front. The fully evolved caudal fluke was very similar in shape to that of lamnid sharks but differed in that the vertebral column passing through it developed a downward kink.
Yet another example of convergent evolution is the cross-fibre structure of the dermis (lower layer of the skin). The body of a large fish can be thought of as a pressurised cylinder, where the hoop stress (the outward circumferential pressure) is twice the stress along the longitudinal axis (Lingham-Soliar 2016). The optimal way of counteracting the stress is to reinforce the skin with fibres aligned at 55° to the axis. Tuna, dolphins and various sharks all have such a network (as do nematode worms!), with angles varying between 25° and 75° depending on where the stress is greatest along the body; the thickest fibres are those deepest in the skin. The fibres are like the warp and weft of a woven fabric. If pulled in the same direction as the fibres, the fabric extends little, but if it is pulled diagonally, it extends a lot. In this way the network allows the body to bend to some degree while still maintaining stiffness. It has been found that the Early Jurassic ichthyosaur Stenopterygius, from southwest Germany’s Posidonia Shale, also possessed such a network. Inevitably, the convergence of different evolutionary lineages upon the same design is ascribed to ‘selection pressures’.
The identity of the land reptile that evolved the ichthyosaur form is not known. Terrestrial animals generally do not get fossilised as often as marine animals, so that may be one reason. Two others are that the founding population was too small for individuals to stand much chance of being fossilised, and the acquisition of marine adaptations was too rapid. When the curtain is pulled back, in the Early Triassic, ichthyosaur fossils are still not abundant. Most of the story has been played out, and most ichthyosaurs are not simply aquatic but fully marine.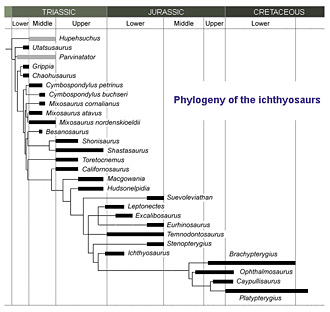 Nonetheless, evolution continued apace, and was fastest at the beginning of the record, over the first ‘10 million years’ (Moon & Stubbs 2020). By the Late Triassic, ichthyosaurs were kings of continental shelf and open ocean alike. As we have noted, the biggest, Shonisaurus, approached the size of a blue whale. It had lost the teeth of its forbears, and like some species of whale, captured its prey by suction.
Nonetheless, evolution continued apace, and was fastest at the beginning of the record, over the first ‘10 million years’ (Moon & Stubbs 2020). By the Late Triassic, ichthyosaurs were kings of continental shelf and open ocean alike. As we have noted, the biggest, Shonisaurus, approached the size of a blue whale. It had lost the teeth of its forbears, and like some species of whale, captured its prey by suction.
Species – early forms especially – continued only a short while before becoming extinct (though new discoveries always have the potential to lengthen their temporal range). A marine mass extinction event at the end of the Late Triassic caused a bottleneck in ichthyosaur evolution. While there was some recovery in diversity (number of species at any one time), disparity (the degree of morphological difference between higher-level groups) dropped permanently and substantially, the final goal of life in the deep seas having been attained. In the Jurassic and Cretaceous ichthyosaurs underwent minor variation, not radical innovation, and were all open-ocean forms.
Diversity waxed and waned as sea temperatures fluctuated. When average temperatures were high, species flourished; few were fossilised in the sedimentary rocks from cooler times. The warmth of sea and land could be the main explanation why reptiles and dinosaurs generally dominated the Mesozoic. Mammals came into their own as temperatures fell. The last ichthyosaur genus passed away during the Cenomanian-Turonian extinction event of the Late Cretaceous, as did the pliosaurs, another group of marine reptiles. By contrast, plesiosaurs and the newly evolved mosasaurs continued to flourish to the end of the period. What caused the extinctions is unclear. It may have been connected with the extrusion of huge volumes of lava in the Pacific, creating the Ontong Lava Plateau.
Did they really evolve from land reptiles? If we look to the fossil record for clues, the answer has to be a resounding ‘No’! … The claim that the ichthyosaurs evolved from terrestrial reptiles simply does not stand up to scientific examination. … Ichthyosaurs did not evolve from anything.
They say the same about the sirenians, when one might determine even from a cursory knowledge of the facts that this is not a tenable view. The evidence for ichthyosaur evolution is clear. The fossils are restricted to the Mesozoic (and particular species restricted to defined periods within the Mesozoic); species become progressively less terrestrial in nature; and, as seen in body sizes, there is a trend early on in their history from near-shore to deep-sea. The more than 80 species of ichthyosaur are not scattered through the fossil record without pattern.

Those who believe that life must have come into being by itself see the evidence that a land reptile evolved into a marine reptile and think, ‘this confirms that all life evolved into existence, without design or purpose.’ The logic is that created species would have had fixed forms; they would have been unable to diversify and adapt themselves to new environments. The appearance of design in the natural world is illusory.
But the argument is nonsense. There is no reason to think that the earth was designed to last forever, changeless and incorruptible. Genetic programs never remain the same. As the world changes, either they adapt and innovate or they face extinction, rather like manufacturers in the business world. The alternative scenario therefore is that the modifications might themselves be programmed, and be orchestrated across the whole genome. Single, accidental mutations in single breeding pairs that, as a consequence, produce more offspring that likewise produce more offspring until the mutation becomes fixed in the whole population are not going to achieve anything significant.
 We have touched on the changes that can be seen or inferred from fossilised bones and skin: the change from foot to flipper as finger bones shortened, became more numerous and more packed together, encased in a streamlined mitten; the streamlining of the whole body, from head to tail; the thickening of the trunk, enabling the diving animal to store more oxygen and body heat; the stiffening of the spinal column; the development of a stiff dorsal fin and tail fluke; the change in eye design, enabling ichthyosaurs to pick out prey in the dark; the acquisition of electro-receptors to help them close in on their prey. The question is, are these changes – to say nothing of the entire preceding history of the lineage – well explained on the supposition that, in the course of genetic miscopying, slight advantages repeatedly passed into the whole population as a result of animals with the mutation leaving more offspring than those without? The history of ichthyosaur DNA cannot be retraced, so we can only consider the question in the light of what seems most likely – likely as judged by the phenomena themselves, not a preconceived belief about the nature of reality. And the phenomena, surely, suggest a co-ordinated sequence of changes directed towards a pre-determined end. The more scientific approach is to attribute large-scale evolutionary change to the intrinsic cause of the organism’s genome, rather than to ‘chance’ entering and leaving the scene like some deus ex machina. Evolution was too fast, even when measured on the conventional timescale.
We have touched on the changes that can be seen or inferred from fossilised bones and skin: the change from foot to flipper as finger bones shortened, became more numerous and more packed together, encased in a streamlined mitten; the streamlining of the whole body, from head to tail; the thickening of the trunk, enabling the diving animal to store more oxygen and body heat; the stiffening of the spinal column; the development of a stiff dorsal fin and tail fluke; the change in eye design, enabling ichthyosaurs to pick out prey in the dark; the acquisition of electro-receptors to help them close in on their prey. The question is, are these changes – to say nothing of the entire preceding history of the lineage – well explained on the supposition that, in the course of genetic miscopying, slight advantages repeatedly passed into the whole population as a result of animals with the mutation leaving more offspring than those without? The history of ichthyosaur DNA cannot be retraced, so we can only consider the question in the light of what seems most likely – likely as judged by the phenomena themselves, not a preconceived belief about the nature of reality. And the phenomena, surely, suggest a co-ordinated sequence of changes directed towards a pre-determined end. The more scientific approach is to attribute large-scale evolutionary change to the intrinsic cause of the organism’s genome, rather than to ‘chance’ entering and leaving the scene like some deus ex machina. Evolution was too fast, even when measured on the conventional timescale.
This sea-going reptile with terrestrial ancestors converged so strongly on fishes that it actually evolved a dorsal fin and tail in just the right place and with just the right hydrological design. The evolution of these forms was all the more remarkable because they evolved from nothing — the ancestral terrestrial reptile had no hump on its back or blade on its tail to act as a precursor.
In the Cenozoic, sea-going mammals with terrestrial ancestors – dolphins and killer whales – also converged ‘on a dorsal fin and tail in just the right place and with just the right hydrological design’. Did these ‘remarkable’ innovations really evolve from nothing? In the mind of a scientist, evolution from nothing should be a scientifically well-founded inference, not a quasi-religious presupposition. Gould’s position is to believe that matter itself is a miracle-worker, the essentially pantheistic view of Epicurus. Without that belief, ‘just the right place’ and ‘just the right hydrological design’ point to processes that were not accidental.
In performing extraordinary feats of adaptation ichthyosaurs were far from alone. Other reptiles that adapted to life at sea include mesosaurs, nothosaurs, pistosaurs, placodonts and thalattosaurs. One branch of the pistosaurs gave rise to the plesiosaurs. Mosasaurs were a further group, originating from varanid lizards in the mid Cretaceous and evolving to become, by the end of the period, ferocious sea monsters up to 10 metres long. They too evolved streamlined forms, flippers, dorsal fins and a caudal fluke, the supporting vertebral column turned down into the lower lobe.
The evolution of reptiles from terrestrial to marine is part of the story of recolonisation following global destruction and extinction before the start of the fossil record. All land animals were wiped out – all but the few pairs saved in the purpose-built, ocean-going lifeboat we know as the ark. It was from them that the ever changing earth was recolonised. The survivors were genetically pre-programmed to adapt to new environments, often in revolutionary ways. While some diapsids were exploring the sea, another group, the pterosaurs, was acquiring the ability to fly, as membranes developed between the digits and transformed their forearms into wings. Certain groups of mammal later underwent similar transformations, becoming whales, dolphins, manatees, seals, otters, moles, bats. One group of birds, the penguins, seemingly gave up the power of flight in return for an ability to swim, in the process colonising the coldest and most hostile continent on Earth.
So far as reptiles and mammals are concerned, the transition was invariably from land to sea – the opposite of the direction taken, in the Darwinian story, when fish became tetrapods. The evolution was horizontal, not vertical: from complex to complex, controlled by signalling and regulatory networks in the genome. Nonetheless, by the end of the Mesozoic most of the marine reptiles had died out. Today only iguanas, snakes and turtles remain from that pioneering age.







